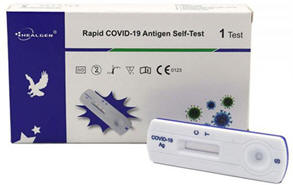
Description
The COVID-19 Tests are in vitro immunochromatographic assay for the rapid, qualitative detection of nucleocapsid protein antigen, IgG/IgM antibodies, neutralizing antibody from SARS-CoV-2 in human Whole blood, serum, plasma, nasopharyngeal swab, nasal swab and oral fluid specimens.
- Key Benefits:Qualitative, visually read results in 10 to 15 minutesDetect COVID-19 Infection with a simple and rapid procedureRoom temperature storage or refrigerated (2-30⁰C / 36-86⁰F)All necessary reagents provided & no equipment needed.High sensitivity and specificity
- Specifications:Clinical Evaluation: Sensitivity 97.25% / Specificity >99% / Accuracy 98.73%Specimen: Nasal swab, Nasopharyngeal swab, Oral Fluid, Whole blood, serum, plasmaTime to Results: 10 to 15 minutesShelf Life: 24 months from the date of manufacture